Hello, welcome to Hangzhou Zhengda Medical Co., Ltd.
NEWS CENTER
PRODUCTS CLASS
Practical Exchange to Deepen Overseas Cooperation
Recently, clients from Japan’s medical industry visited Hangzhou and toured the facilities of Hangzhou Zhengda Medical Equipment Co., Ltd. to conduct on-site inspections and business negotiations. Company
executives and relevant personnel from production and R&D accompanied the delegation throughout the visit, providing a comprehensive overview of the company’s production operations, technological R&D,
and product quality control. Both parties engaged in in-depth discussions regarding the details of potential cooperation, laying the groundwork for future international collaboration.
I. Deepening Industry Expertise, Strengthening the Foundation of Quality
Hangzhou Zhengda Medical Equipment Co., Ltd. has been deeply rooted in the medical equipment sector for over two decades. Adhering to the business philosophy of “Quality First, Integrity and Mutual Benefit,”
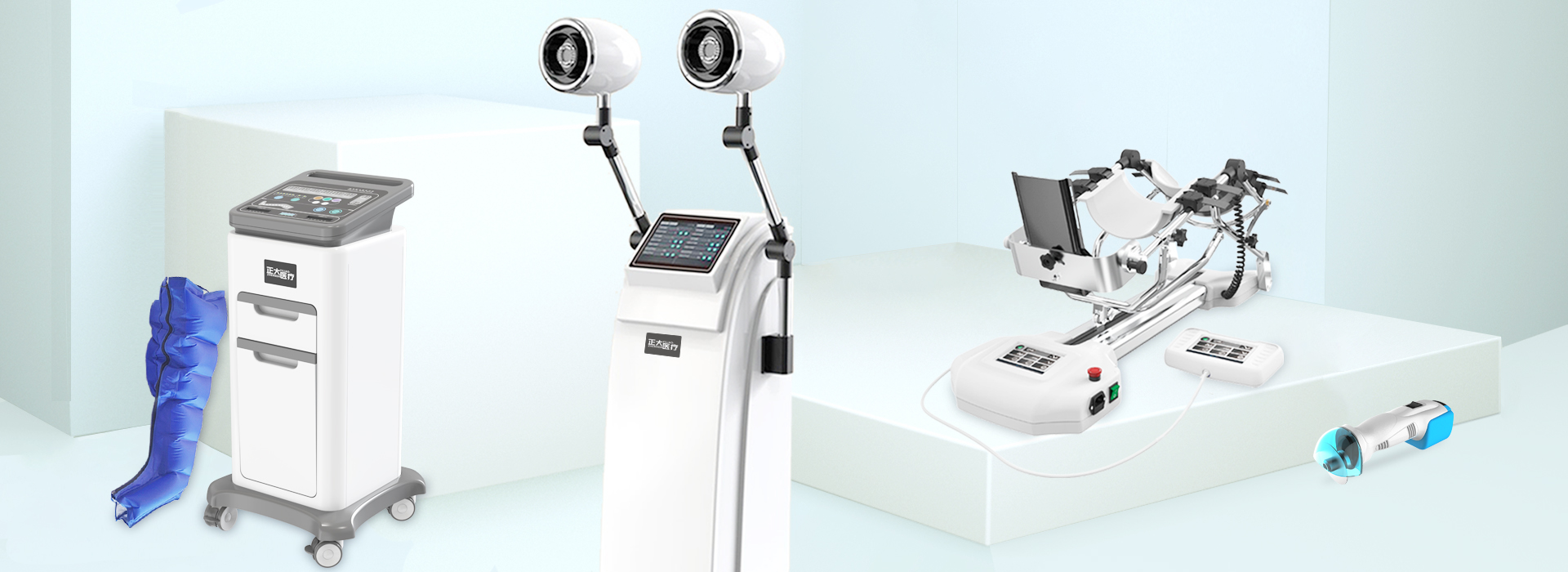
the company primarily engages in the R&D, production, and sales of equipment for orthopedics, rehabilitation, and operating rooms. Currently, the company’s product portfolio includes joint rehabilitation devices,
electric pneumatic hemostats, cold air therapy units, shockwave therapy units, medical plaster saws, and fumigation therapy units, among other series. These products are widely used in clinical settings across
medical institutions of all levels.
II. On-Site Visit: Recognizing Quality and Capabilities
During the visit, the Japanese clients toured the company’s product showroom, standardized production workshops, and quality testing center, observing firsthand every stage of product R&D, manufacturing,
and quality control. In the product showroom, the clients inquired in detail about the technical specifications, clinical efficacy, and usage precautions of various core products. They also personally tested the
equipment, expressing their approval of the products’ user-friendly design, manufacturing craftsmanship, and operational stability.
In the production workshops and quality testing center, the clients focused on understanding the operational status of production equipment, standardized work procedures, and the end-to-end quality traceability
system. They expressed recognition of the company’s standardized production and quality management model, which strictly adheres to the ISO 13485 quality management system and features multiple products
certified by CE and FDA.
III. Roundtable Discussion: Exploring Collaboration Opportunities
Following the tour, both parties held a roundtable discussion to engage in practical discussions on specific matters such as overseas market demand, product adaptation and optimization, long-term supply
guarantees, and future cooperation models. Drawing on the actual characteristics of the local medical market, the Japanese client offered targeted suggestions regarding the adaptability of the company’s
products. The company’s relevant officials addressed each of the client’s concerns and stated that they would further optimize product details based on client needs, ensure the stability of product supply, and
provide comprehensive after-sales service.
Both parties agreed that the medical industries of China and Japan are highly complementary and possess significant potential for cooperation. This visit established a solid communication platform and laid the
groundwork for a preliminary consensus on jointly exploring international markets in the future.
The Japanese clients’ visit served as an on-site evaluation and affirmation of Hangzhou Zhengda Medical Devices Co., Ltd.’s product quality, production capabilities, and service standards, marking a significant
step forward in the company’s efforts to advance its international operations and expand into overseas markets. Moving forward, the company will remain committed to its founding principle of quality, deepen its
focus on technological innovation, continuously enhance product competitiveness, actively align with overseas market demands, strive for mutual benefit and win-win outcomes with global clients, and contribute
to the development of the healthcare industry.